Minerals are the fundamental building blocks of Earth’s crust, and their unique crystal structures define their physical and chemical properties. The way atoms arrange themselves within a mineral determines its shape, hardness, cleavage, and even its color. Understanding crystal structures in mineralogy helps geologists and material scientists classify minerals, predict their behavior, and explore their practical applications.
In this article, we will explore how minerals form their distinctive crystal structures, the different types of crystal systems, and why these structures matter in the field of mineralogy.
What Are Crystal Structures?
A crystal structure is the ordered arrangement of atoms, ions, or molecules in a mineral. This internal framework influences the mineral’s external appearance, forming characteristic geometric shapes known as crystal habits. The repetition of atomic patterns on a microscopic scale results in the macroscopic crystal forms observed in nature.
Crystal structures develop under specific temperature and pressure conditions, and slight variations can lead to the formation of polymorphs—minerals with the same chemical composition but different crystal structures (e.g., diamond and graphite).
The Seven Crystal Systems
Minerals naturally form in seven distinct crystal systems, classified based on their symmetry, axis lengths, and angles. Each system influences the way a mineral grows and interacts with light and other forces.
1. Cubic (Isometric) System
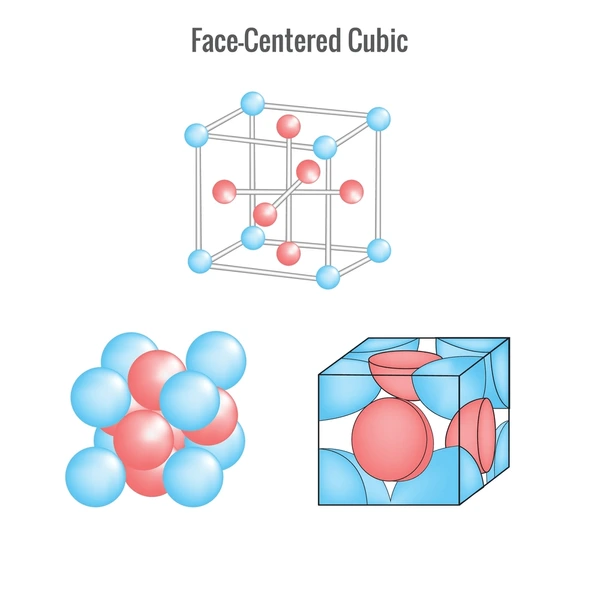
The cubic system is characterized by equal-length axes that intersect at right angles. Minerals in this system tend to form symmetrical, block-like shapes such as cubes or octahedrons.
Examples:
- Pyrite (FeS₂): Known as “fool’s gold,” it often forms perfect cubic crystals.
- Halite (NaCl): Table salt, which naturally crystallizes in cubic formations.
- Diamond (C): One of the hardest substances, with a cubic atomic structure.
2. Tetragonal System
Similar to the cubic system, but one axis is longer or shorter than the other two. This results in elongated crystal forms.
Examples:
- Zircon (ZrSiO₄): A durable mineral used in dating ancient rocks.
- Rutile (TiO₂): A common mineral in metamorphic rocks.
3. Hexagonal System
This system features three equal horizontal axes at 120° angles, with a vertical axis perpendicular to them. It produces six-sided crystal formations.
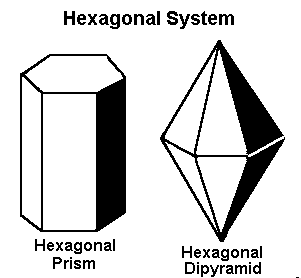
Examples:
- Quartz (SiO₂): One of the most abundant minerals, forming hexagonal prisms.
- Beryl (Be₃Al₂Si₆O₁₈): The mineral group that includes emerald and aquamarine.
4. Trigonal System
A subset of the hexagonal system, but with a three-fold rotational symmetry instead of six. Many well-known minerals belong to this system.
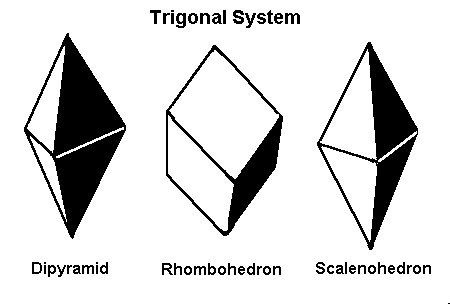
Examples:
- Calcite (CaCO₃): Often forms rhombohedral or prismatic crystals.
- Tourmaline: Displays elongated, prismatic forms with unique color variations.
5. Orthorhombic System
In this system, all three axes have different lengths but intersect at right angles. It results in prismatic and tabular crystal shapes.
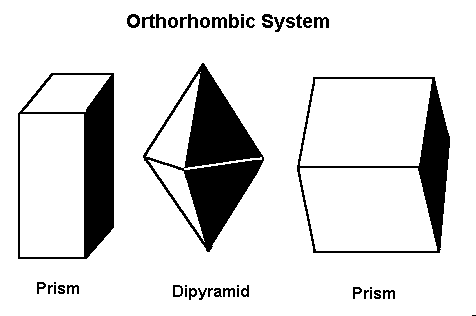
Examples:
- Topaz (Al₂SiO₄(F,OH)₂): Often forms elongated, columnar crystals.
- Sulfur (S): Bright yellow, brittle crystals found in volcanic regions.
6. Monoclinic System
The monoclinic system has three unequal axes, with one inclined at an oblique angle. This leads to asymmetrical crystal forms.
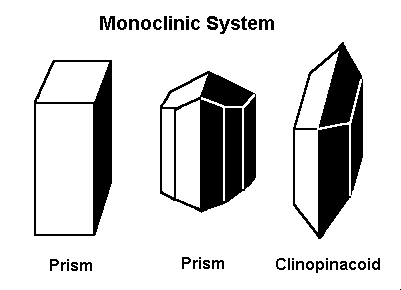
Examples:
- Gypsum (CaSO₄·2H₂O): A soft mineral that forms elongated crystals.
- Augite: A common dark-colored pyroxene found in igneous rocks.
7. Triclinic System
The least symmetrical system, where all three axes are of different lengths and none meet at right angles. Triclinic minerals tend to have irregular and distorted crystal forms.

Examples:
- Microcline (KAlSi₃O₈): A type of feldspar often found in granite.
- Kyanite (Al₂SiO₅): Forms bladed crystals with a unique blue hue.
Why Do Crystal Structures Matter?
The internal arrangement of atoms in a mineral determines several key physical properties, including:
- Hardness: The strength of atomic bonds defines a mineral’s resistance to scratching (Mohs hardness scale).
- Cleavage and Fracture: Minerals break along specific planes based on their atomic structure.
- Optical Properties: How a mineral interacts with light depends on its crystal structure.
- Stability and Weathering: Some structures are more resistant to heat, pressure, and chemical changes.
For example, diamond and graphite both consist of carbon but differ in structure—diamond’s cubic lattice makes it the hardest known mineral, while graphite’s layered hexagonal structure allows it to be soft and slippery.
The Role of Crystal Structures in Industry and Science
Understanding crystal structures has practical applications in geology, material science, and industry:

- Gemology: Identifying and grading gemstones based on crystal form.
- Mining and Resource Exploration: Locating valuable mineral deposits by studying crystal habits.
- Engineering and Technology: Using mineral properties in semiconductors, ceramics, and synthetic materials.
- Pharmaceuticals: Designing drugs based on molecular crystal structures.
Final Thoughts
Crystal structures are the foundation of mineralogy, determining a mineral’s shape, strength, and behavior. From the perfect cubes of pyrite to the hexagonal prisms of quartz, the atomic arrangements within minerals influence everything from geological formations to industrial applications.
By studying these structures, geologists and scientists unlock a deeper understanding of Earth’s materials, paving the way for innovations in technology, resource exploration, and environmental science. Whether admiring a natural quartz crystal or analyzing a diamond’s brilliance, crystal structures reveal the intricate beauty and complexity of the natural world.

